Frequently Asked Questions
- What is waste heat?
- What are typical uses for waste heat?
- How does waste heat to power work?
- Where is the Opportunity for Waste Heat to Power?
- What are major sources of industrial waste heat?
- Where is Echogen located?
- How many patents has Echogen filed?
- Does Echogen do anything other than Waste Heat to Power?
- Where did Echogen’s solution originate?
- What is Supercritical CO2?
What is waste heat?
The term “waste heat,” refers to the thermal energy produced by machines, electrical equipment, and industrial processes that escapes into the environment unused. Often this waste heat can be recovered and used for other purposes.
What are typical uses for waste heat?
The most common uses for waste heat are:
- Recycling heat back into the existing manufacturing process
- Transferring the waste heat from one process to another within the facility
- Converting heat into electrical (or mechanical) power [Waste Heat to Power (WHP) or Waste heat to Mechanical Drive (WH2MD)]
The first two options are generally economical, but require a local use for the recovered heat. The third option creates a product, electrical power, which can be consumed onsite or is easily transmitted and has high economic value.
How does waste heat to power work?
Waste Heat to Power (WHP) creates electricity by heating a fluid at high pressure, then expanding the fluid through a turbine to power an electric generator. In a classic steam Rankine waste heat system, the turbine is turned by a pressurized water vapor (steam), which has been boiled by the waste heat stream. The steam is then cooled off and condensed to liquid water, pumped back to high pressure, and begins the process again. Most WHP systems today are adaptations of the original Rankine design – a system that takes heat from an external source and converts it to power (work) through a closed-loop thermodynamic system.
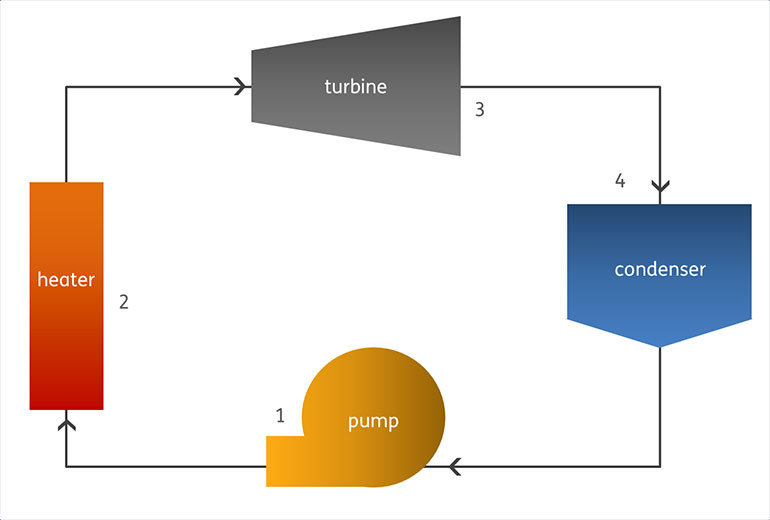
- Step 1: Cooled water leaving the condenser is pumped to the boiler.
- Step 2: The waste heat source heats the water in the boiler, causing the water to boil and become pressurized steam.
- Step 3: The pressurized steam is injected into the turbine, which causes the turbine blades to spin. The turbine is connected to a generator, which creates electricity.
- Step 4: The steam leaving the turbine is sent to the condenser to convert back to liquid water before the cycle begins again.
Where is the Opportunity for Waste Heat to Power?
The greatest potential source for WHP generation is in the industrial sector. In 2009, this sector consumed the largest amount of energy in the United States, accounting for more than 30 percent of all domestic energy consumption . Roughly one-third of this energy consumed by industry is discharged as thermal losses due to inefficiencies.
In the United States alone, it is currently estimated that 6,000 to 8,000 megawatts [MW] of electric generating capacity is available for recovery from industrial operations per year. Non-industrial applications, including exhaust from natural gas pipeline compressor drives and landfill gas engines, represent another 1,000 to 2,000 MW of power capacity, for a total of 7 to 10 gigawatts (source).
What are major sources of industrial waste heat?
Energy intensive processes—such as those occurring at refineries, steel mills, glass furnaces, and cement kilns—all release hot exhaust gases and waste streams that can be harnessed by a heat engine to generate electricity.
Some specific operations that create valuable, high-temperature waste heat are:
- Glass manufacturing:
- melting furnaces, annealing ovens, and tempering furnaces
- Cement milling:
- Mining and quarrying, crushing, clinker production in kilns, cement milling, rotary cement kilns using coal or natural gas
- Steel manufacturing:
- coke ovens, blast furnaces, reheat furnaces, basic oxygen, and electric arc furnaces
- Petroleum refining:
- petroleum coke calciners
- Chemical processing:
- thermal oxidizers
Other operations that generate valuable waste heat include gas turbine-driven compressors at natural gas compressor stations, natural gas turbines and reciprocating internal combustion engines or turbines at landfills, and flared energy sources at Oil and Gas Production facilities.
Where is Echogen located?
Country: United States
State: Ohio
City: Akron
We are located about 40 miles south of Cleveland, in downtown Akron, Ohio. Our headquarters are in the former "Hamlin Steel" building.
How many patents has Echogen filed?
Echogen currently has 5 issued patents; and 42 patents pending. Echogen’s commitment to innovation is an integral part of our workplace.
Does Echogen do anything other than Waste Heat to Power?
Echogen’s engine is a platform technology that has potential far-reaching applications. We are currently funded under the DOE’s SunShot program to develop a solar-powered system that also uses sCO2 as a working fluid. Our work with the University of Texas and Lawrence Berkeley National Laboratory studying geothermal energy and carbon sequestration continues to yield new discoveries for our technology to help our environment. We are committed to researching new ways for our technology to benefit more industries and the planet.
Where did Echogen’s solution originate?
Echogen’s founders originally licensed a NASA technology, the “Champagne” CO2 absorption heat pump, and developed further intellectual property and patent-pending applications that extended the heat pump technology to a WHP engine.
Echogen’s first prototype, completed in 2007, was a version of the “Champagne” absorption heat pump using carbon dioxide and a preferred secondary fluid. Echogen then converted the heat pump to a WHP engine, reducing to practice a first approach to the power generation cycle. A second prototype system, completed in early 2009, used pure carbon dioxide and proved that a transcritical cycle heat engine could be built to produce electricity from waste heat for commercial applications, and established a development pathway for first production units.
What is Supercritical CO2?
Supercritical carbon dioxide (sCO2) is a fluid state of carbon dioxide when it exceeds its critical temperature and critical pressure. In this state, it assumes properties intermediate between those of a gas and a liquid, and in fact loses the distinction between gas and liquid phases. Its high density manifests itself in an equipment set with a very small footprint. sCO2 is a non-flammable, non-toxic, non-corrosive, thermally stable working fluid. Because of these properties, sCO2 can interact more directly with the heat source, eliminating the need for a secondary thermal loop, and further reducing the total installed cost of the system.
Properties of supercritical CO2 allow for:
- Flexible cycle - can integrate with wide variety of exhaust temperatures from low-speed diesels to simple cycle gas turbines
- Compact, closed-loop system with minimal operating & maintenance support
- Competitive thermal-to-electric power conversion efficiency with typically lower capital cost vs. steam or ORC technologies